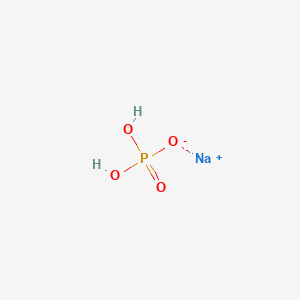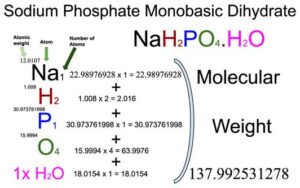
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes
![Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 5 Kilograms: Amazon.com: Industrial & Scientific Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 5 Kilograms: Amazon.com: Industrial & Scientific](https://m.media-amazon.com/images/I/71N1WwL3FIL.jpg)
Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 5 Kilograms: Amazon.com: Industrial & Scientific
![Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific](https://m.media-amazon.com/images/I/41zZzkwboAL._SR600%2C315_PIWhiteStrip%2CBottomLeft%2C0%2C35_SCLZZZZZZZ_FMpng_BG255%2C255%2C255.jpg)
Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%20500G.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs
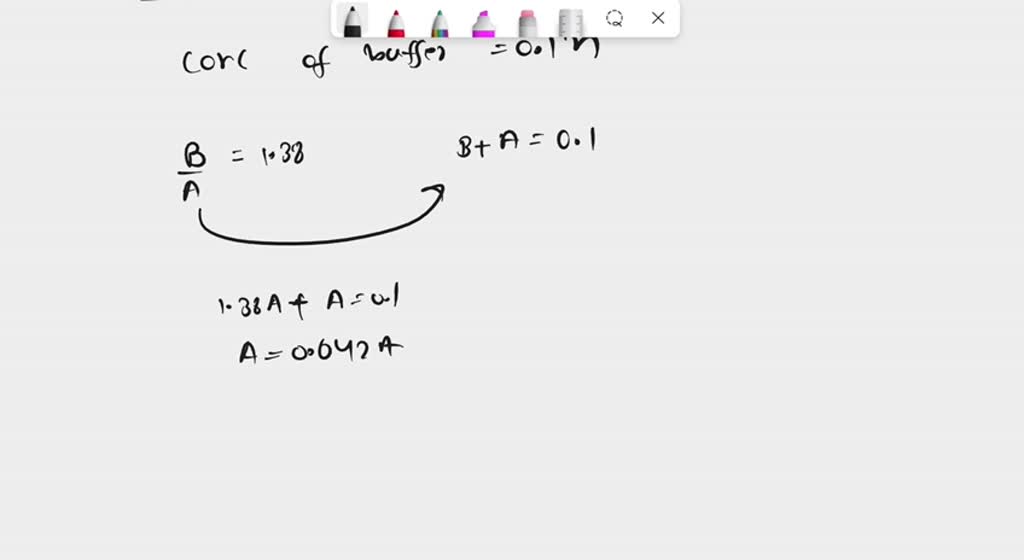
SOLVED: 1. How many g of Na2HPO4 and NaH2PO4 2H2O would you need to prepare 1L of 0.1M sodium phosphate buffer pH 7.0? (Hint= use the Henderson-Hasselbalch equation) Express your answer to

SOLVED: What is the balanced equation and the net ionic equation. NaH2PO4 + Na2HPO4 + H2O + MgCl2 ⇒ NaH2PO4 + Na2HPO4 + H2O + NaCl ⇒ ( we did an experiment
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/Tris%20500g.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs
![S23120-1000.0 - Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen phosphate monohydrate], 1 Kilogram S23120-1000.0 - Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen phosphate monohydrate], 1 Kilogram](https://d2gdaxkudte5p.cloudfront.net/system/images/plabel_14789_20220901-031402.jpg)
S23120-1000.0 - Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen phosphate monohydrate], 1 Kilogram
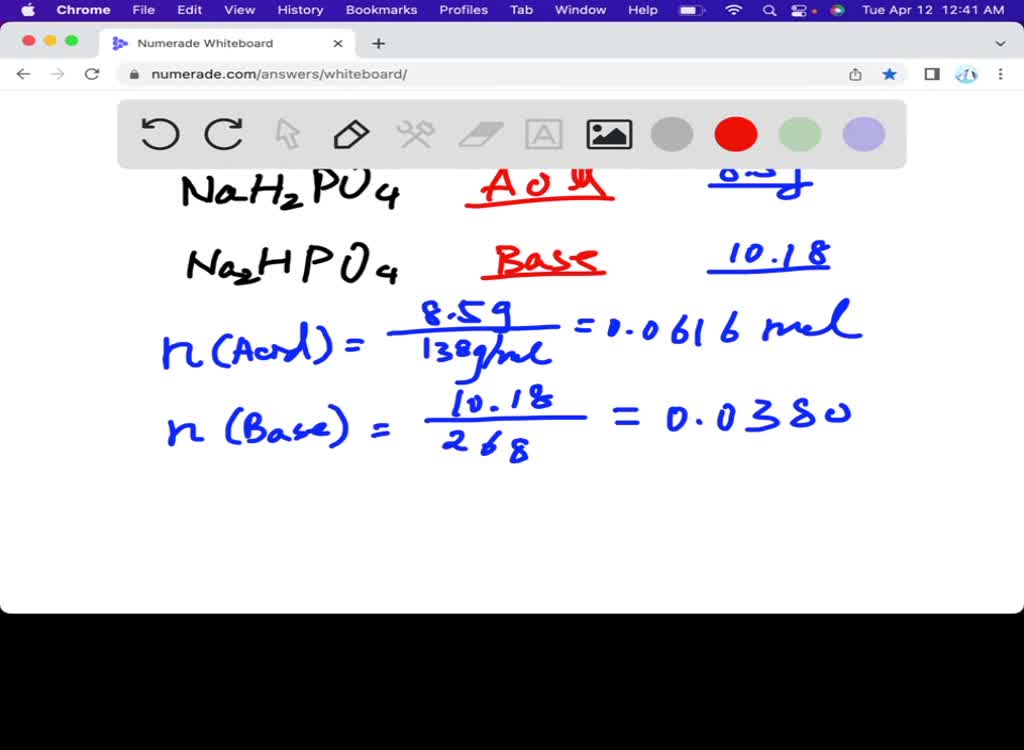
SOLVED: Calculate the mass of NaH2PO4 ∙ H2O (in grams) required to prepare 250mL of pH 7.21 buffer solution if the total buffer concentration is to be 0.20M. The Ka of NaH2PO4∙H2O

The equivalent weight of NaH2PO4 in the reaction NaH2PO4+KOH→NaKHPO4 + H2O (Given Atomic masses: Na = 23. K = 39, P = 31)

10049-21-5, 137.99, Sodium Phosphate, Monobasic, Monohydrate, Crystal, Reagent, ACS - 6NNX7|S1395-2.5KG - Grainger