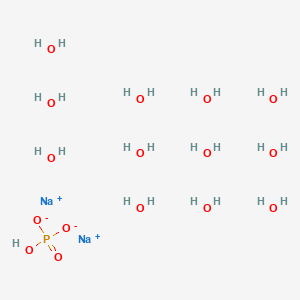
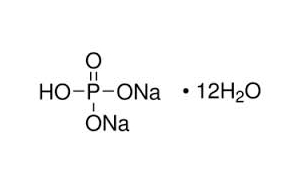
Jual diSodium hidrogen fosfat dodekahidrat Na2HPO4.12H2O eceran - Kota Malang - Phy Edumedia | Tokopedia

Na2hpo4.12h2o 357.98 Molecular Weight Disodium Hydrogen Phosphate - Buy Molecular Weight Disodium Hydrogen Phosphate,Na2hpo4.12h2o,Disodium Salt Product on Alibaba.com

Enhancing the Heat Storage Performance of a Na2HPO4·12H2O System via Introducing Multiwalled Carbon Nanotubes | ACS Omega
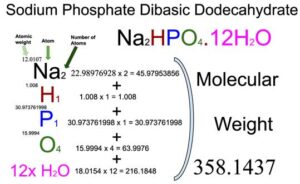
Sodium Phosphate Dibasic Dodecahydrate (Na2HPO4.12H2O) Molecular Weight Calculation - Laboratory Notes
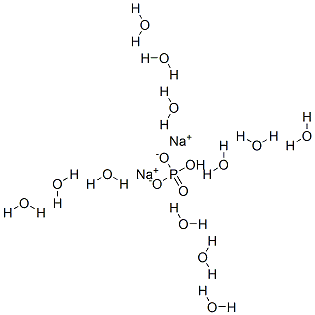
Sodium hydrogen phosphate dodecahydrate, China Sodium hydrogen phosphate dodecahydrate, Sodium hydrogen phosphate dodecahydrate exporters, China Sodium hydrogen phosphate dodecahydrate Suppliers - zxphosphates

Construction of Na2CO3·10H2O-Na2HPO4·12H2O eutectic hydrated salt/NiCo2O4-expanded graphite multidimensional phase change material - ScienceDirect
![SOLVED: 1.) calculate how many grams of disodium hydrogen phosphate dodecahydrate [Na2HPO4 * 12H2O] ans water should be used to prepare 27 grams of 4.5 % solution of this salt. 2.) How SOLVED: 1.) calculate how many grams of disodium hydrogen phosphate dodecahydrate [Na2HPO4 * 12H2O] ans water should be used to prepare 27 grams of 4.5 % solution of this salt. 2.) How](https://cdn.numerade.com/ask_previews/d3e9cad5-07e7-4af0-b983-9e3d75ec767e_large.jpg)
SOLVED: 1.) calculate how many grams of disodium hydrogen phosphate dodecahydrate [Na2HPO4 * 12H2O] ans water should be used to prepare 27 grams of 4.5 % solution of this salt. 2.) How