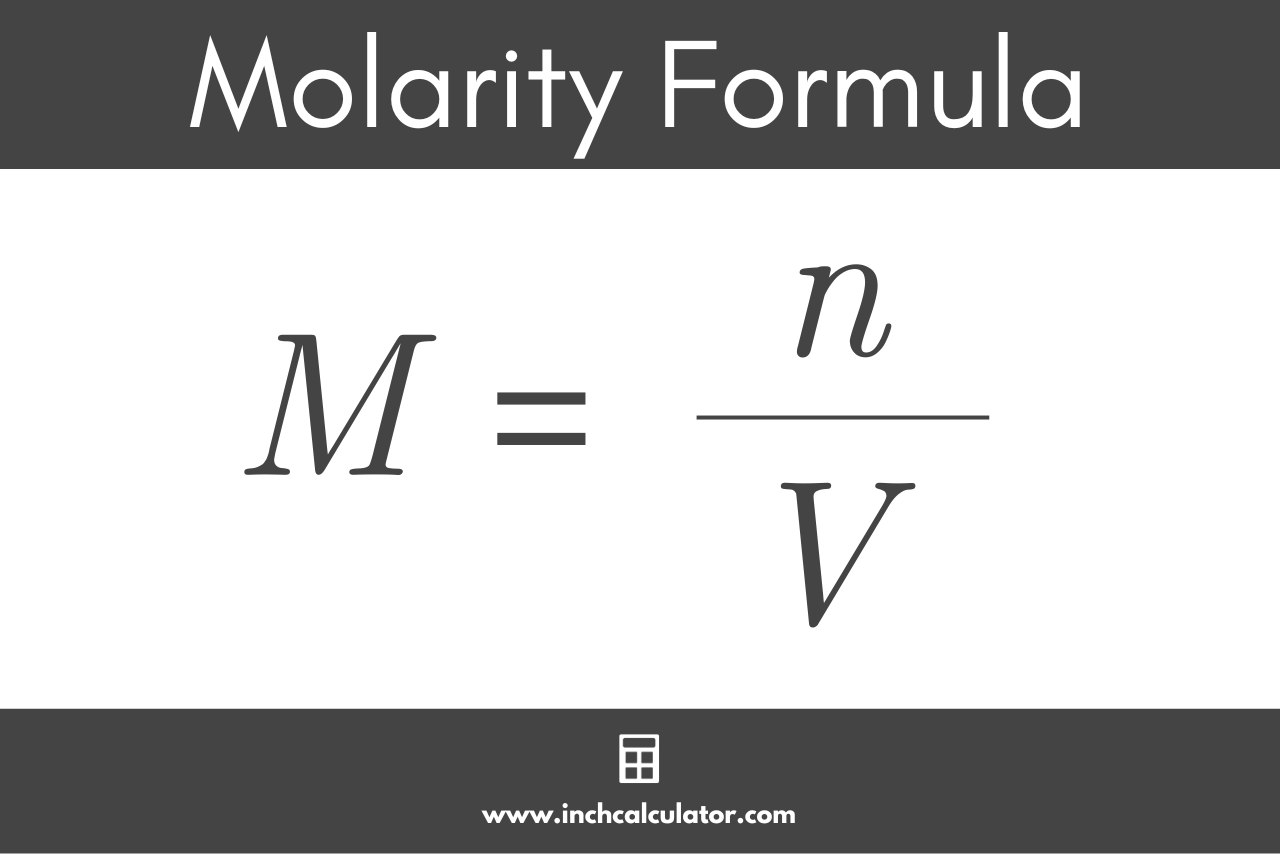![SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6 SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6](https://cdn.numerade.com/ask_previews/1a8be9cb-b10d-490e-9333-ae6f4d305d94_large.jpg)
SOLVED: What volume (in mL) of a 2.40 mol L −1 magnesium bromide solution would contain 3.40 x 10^23 bromide [Br− (aq)] ions? A) 118 B) 12.3 C) 543 D) 58.9 E) 74.6

Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download
What is H+in mol/L of a solution that is 0.20 M CH3COONa and 0.10 M in CH3COOH? (Ka for CH3COOH =1.8x10 5)

Calculate the volume of 1.00 mol ${{L}^{-1}}$ aqueous sodium hydroxide that is neutralised by 200 mL of 2.00 mol ${{L}^{-1}}$ aqueous hydrochloric acid arid the mass of sodium chloride produced - CBSE

PPT - MOLARITY A measurement of the concentration of a solution PowerPoint Presentation - ID:5814201

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = mol / L Calculate. - ppt download