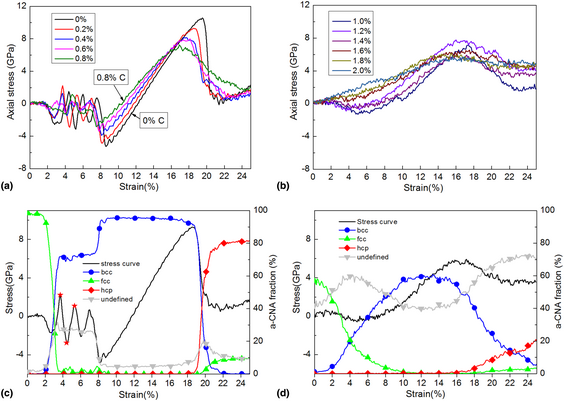
The strain-induced martensitic phase transformation of Fe–C alloys considering C addition: A molecular dynamics study | Journal of Materials Research | Cambridge Core

Ferrocene. Molecular model of the organometallic compound ferrocene (C10.H10.Fe). This is a metallocene consisting of two cyclopentadienyl rings bound... - SuperStock
Ferrocene. Molecular model of the organometallic compound ferrocene (C10.H10.Fe). This is a metallocene consisting of two cyclopentadienyl rings bound by a central iron atom. The discovery of this compound and its properties

Fe(deferasirox)2: An Iron(III)-Based Magnetic Resonance Imaging T1 Contrast Agent Endowed with Remarkable Molecular and Functional Characteristics | Journal of the American Chemical Society
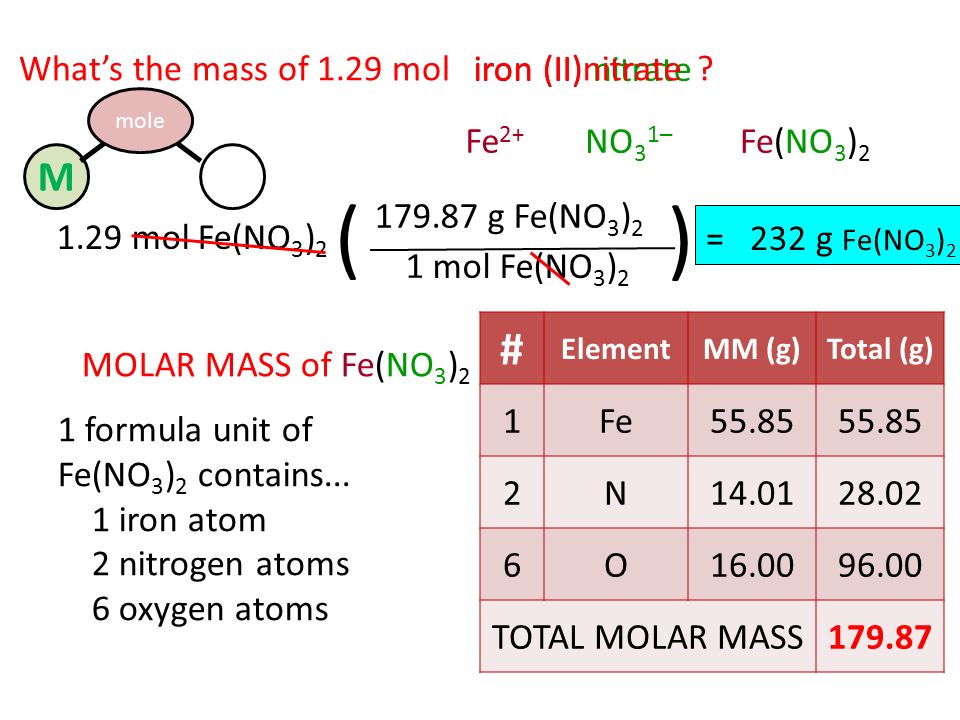
MOLE (mol) Mass (g) Particles (atoms,m'c or f.u.) Which conversion factors that allow you to convert from mass to moles and from particles to moles. - ppt download
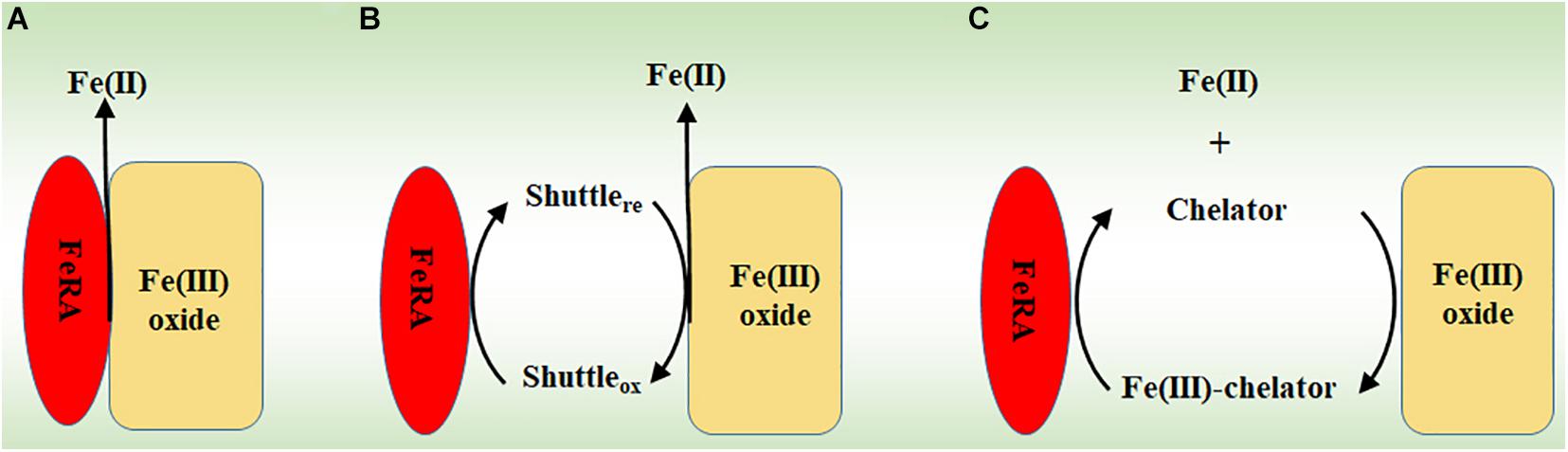
Frontiers | The Proposed Molecular Mechanisms Used by Archaea for Fe(III) Reduction and Fe(II) Oxidation
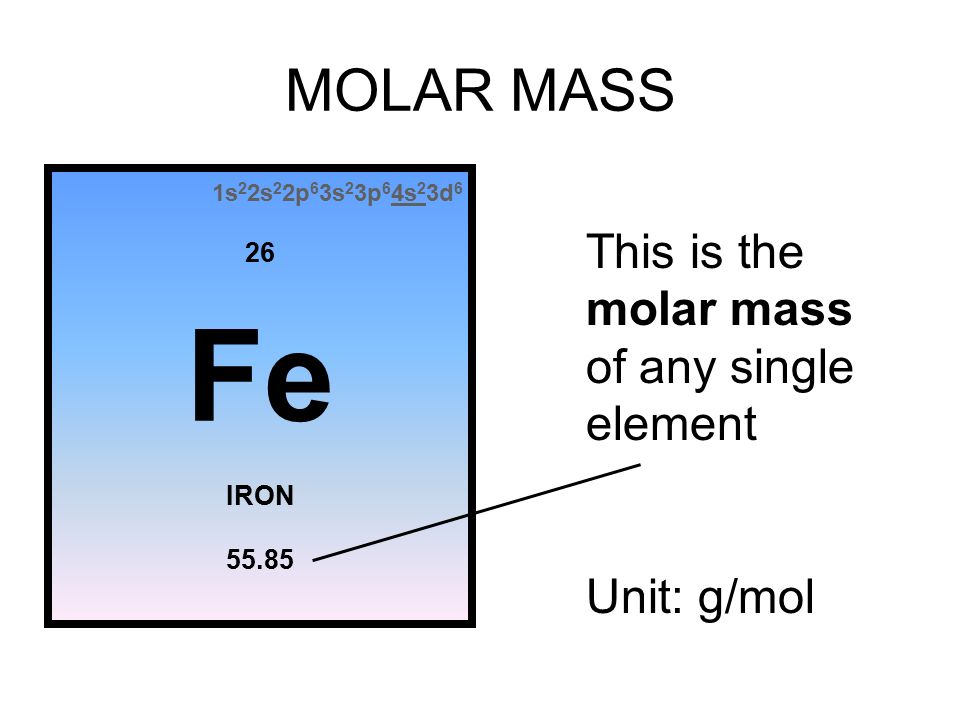





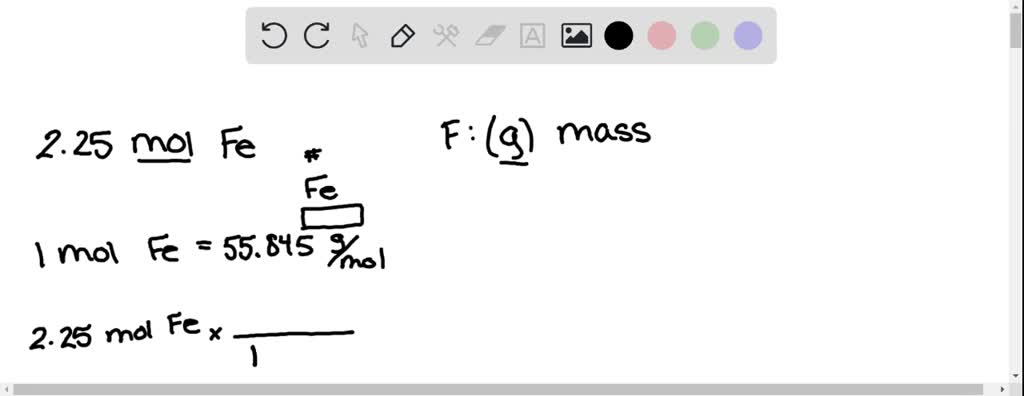
![ANSWERED] Calculate the molar mass of Fe 3(PO 4) 2... - Inorganic Chemistry ANSWERED] Calculate the molar mass of Fe 3(PO 4) 2... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/63334580-1657459641.4436913.jpeg)