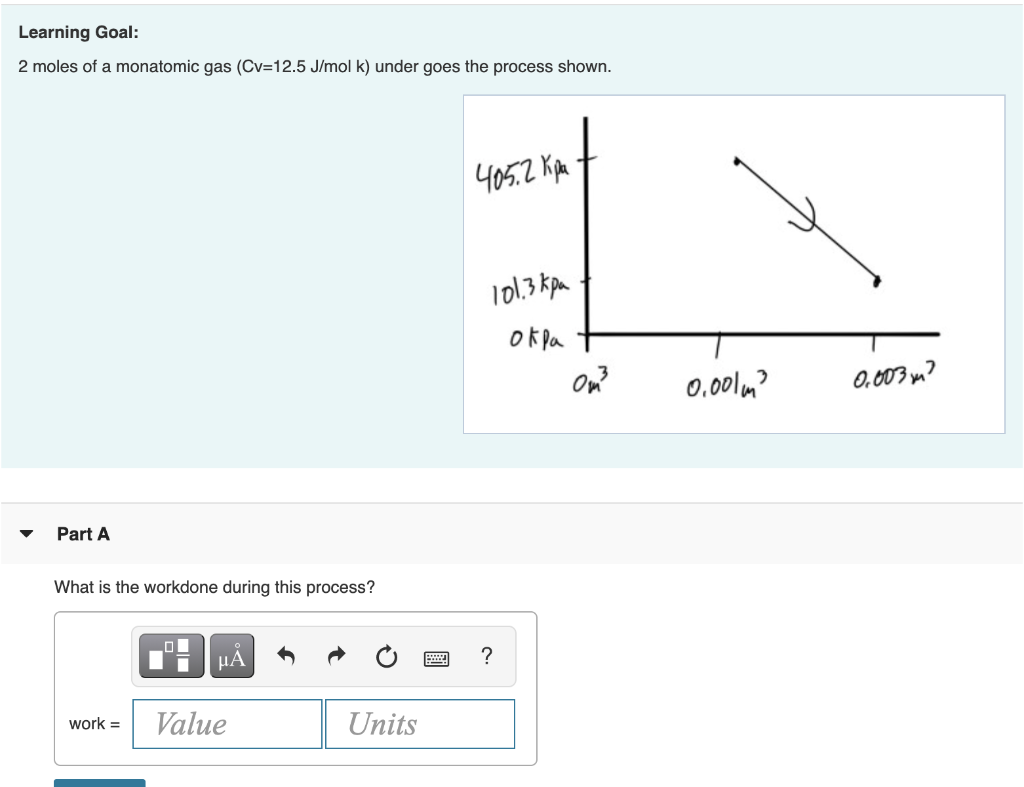
If Cp=20J/mol.K then for 1mole gas we get Cv=.....? You can use Cp/Cv=1.7_-_ #physics #problems - YouTube
A vessel contains 1mol of oxygen and 2 mol of helium. What is the value of CP/CV of the mixture? - Quora
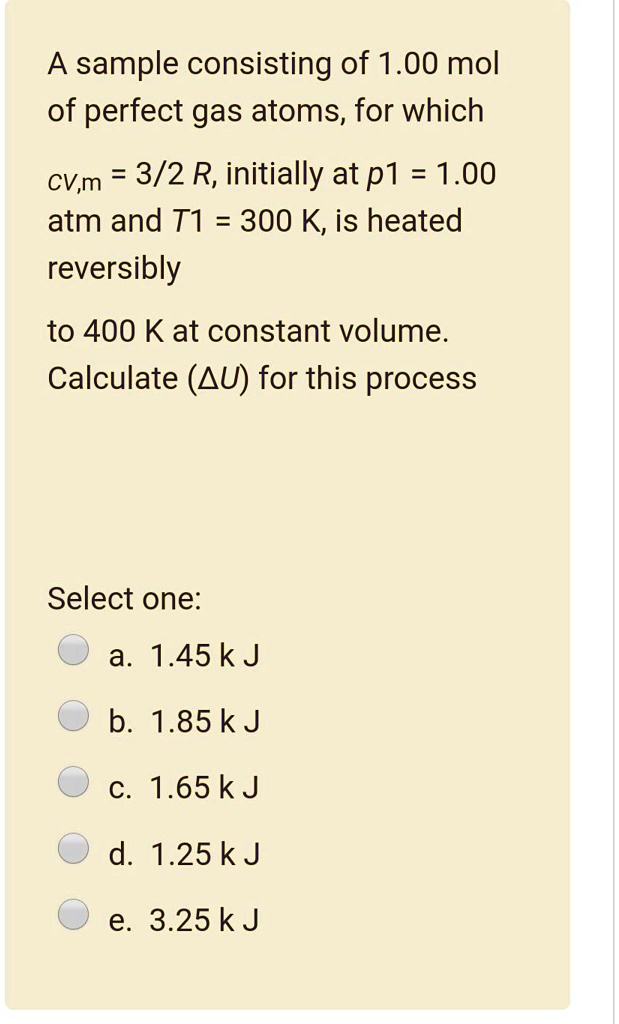
SOLVED: A sample consisting of 1.00 mol of perfect gas atoms, for which CVm = 3/2 R, initially at p1 1.00 atm and T1 = 300 K, is heated reversibly to 400
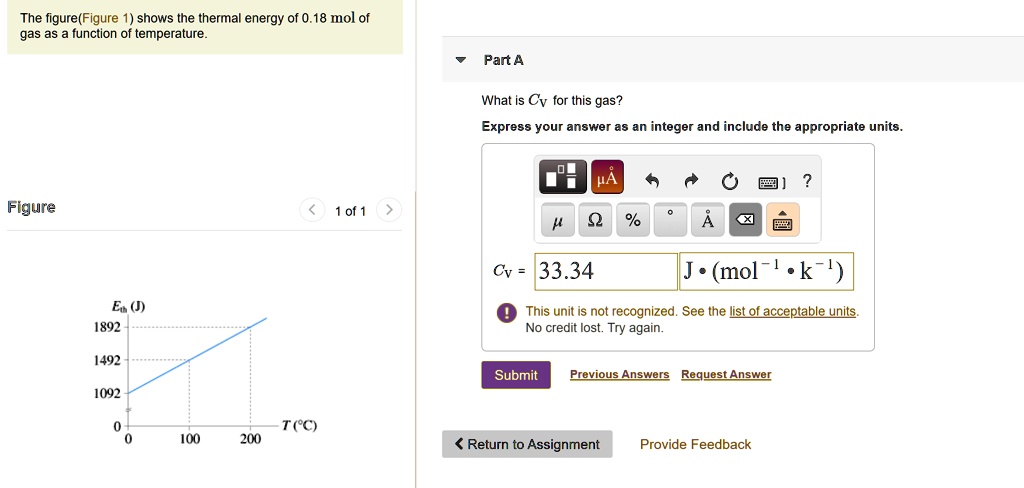
SOLVED: The figure(Figure shows the thermal energy of 0.18 mol of gas as function of temperature Part A What is Cv for this gas? Express your answer as an integer and include
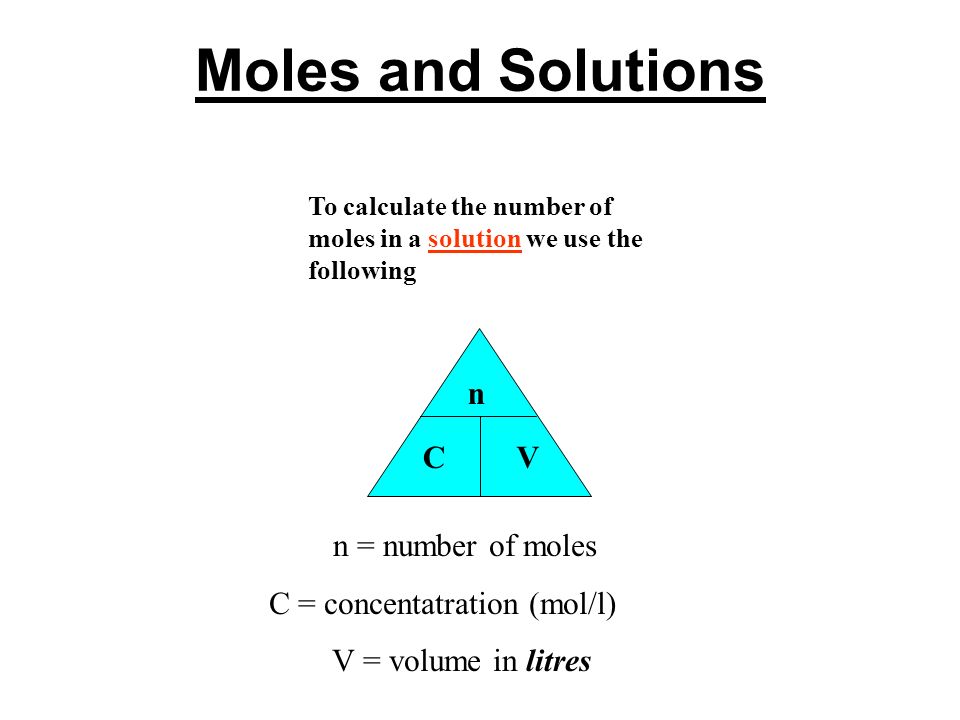
Moles and Solutions g n gfm To calculate the number of moles in a solution we use the following n CV n = number of moles C = concentatration (mol/l) V. - ppt download