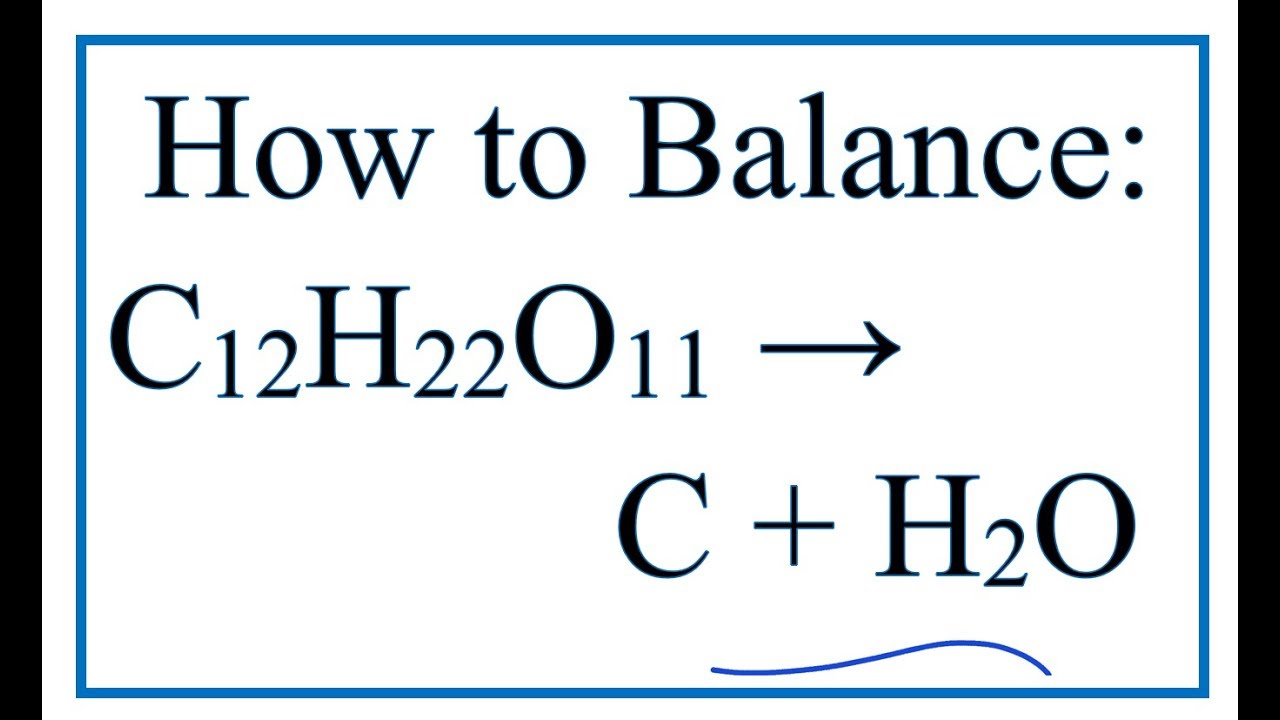
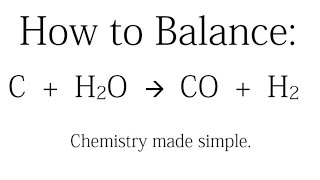SOLVED: A mixture of 0.466 M H2O , 0.481 M Cl2O , and 0.718 M HClO are enclosed in a vessel at 25°C . H2O(g)+Cl2O(g)↽−−⇀2HOCl(g)𝐾c=0.0900 at 25°C Calculate the equilibrium concentrations of

Use the problem below to answer the question: 34 grams of carbon reacted with an unlimited amount of H20. - Brainly.com

SOLVED: How many grams of H2 would be formed if 34 grams of carbon reacted with an unlimited amount of H2O? The reaction is C + H2O → CO + H2 The